 |
|
|
|
|


Developing new treatments for people suffering from sleeping sickness (HAT) or visceral leishmaniasis (VL) is a long and difficult process, especially when clinical trials are carried out in rural areas in Africa where health infrastructure is limited or non-existent. However, it is of critical importance to conduct clinical trials in such areas so as to ensure that the treatments being tested will actually meet the real-life needs of the patients.
Down-to-earth challenges
Areas with a high incidence of sleeping sickness and VL are mostly in remote settings where patients have limited access to health care and skilled staff, and in some cases, are exposed to a general political instability.
In very practical terms, patients may walk for 5 days to reach a hospital in order to receive treatment. This means that by the time patients reach help they may be very sick. Patients often suffer from malnourishment and co-infections such as malaria, pneumonia, or HIV. Patients may or may not receive a correct diagnosis due to lack of adequate, field-adapted tools and trained health professionals. Thus, sleeping sickness might be misidentified as VL, or VL might be misdiagnosed as malaria.
In respect to clinical trials, several major challenges can be identified as study sponsors endeavour to abide by international Good Clinical Practice (GCP) standards:
- In some countries the approval process to start a clinical trial may take over a year (from first submission to approval for recruitment of the first patient) as administrative and regulatory processes must be properly followed in order to receive approval to conduct clinical trials.
- Conducting clinical trials depends on the participation and consent of several hundreds of HAT or VL patients who are located in remote areas with poor resources.
- In order for patients to participate in a clinical trial, the diagnosis must be confirmed through invasive procedures such as lumbar punctures or splenic aspirates necessary to detect the parasite as more readily field-adapted diagnostics are not yet available.
- Ensuring patient compliance and follow-up can also be difficult. For both diseases, treatment is long and must be given at a health centre; the inconvenience of trial periods lasting up to 30 days can have a tangible impact on the family. As in the case of VL, half the patients diagnosed with this disease are children. A family member will usually stay in the vicinity of the treatment centre for the duration, meaning someone has to care and provide for the siblings back at home. The difficulty will be even further compounded due to the possible loss of wages.
In the case of HAT, follow-up visits are required up to 18 months post-treatment to ensure that the treatment has effectively killed the parasite. If patients forget their appointments or are unable to come, health care workers are obliged to seek these people out, often necessitating a trip to the patient's village far away from the treatment centre. - There is often limited experience in ethical review of clinical trials in some of the endemic countries where trials are being conducted.
- Armed conflict and political instability are also important elements to take into consideration. Apart from the obvious impact on the local populations, such situations add further complexities and could jeopardise the outcome of the clinical trial. Take for instance the recent political instability and subsequent violence in Kenya this resulted in an approximate 3-month delay for the 2 clinical trials DNDi was conducting in Western Kenya.
The Partner advantage
To address these challenges, DNDi has facilitated the formation of LEAP and HAT Platforms, which are comprised of a group of scientists, academic institutions and representatives from Ministries of Health from disease endemic countries. Acting together along with MSF operational efforts is the best way to develop clinical trial capacity to ensure centres of excellence for both the trials and the treatment of patients. Some key activities of these platforms include addressing the issues described above: developing appropriate clinical trial methodologies, overcoming system challenges, strengthening clinical trial capacity both in terms of infrastructure and human resources, sharing information, and facilitating communication among all involved.
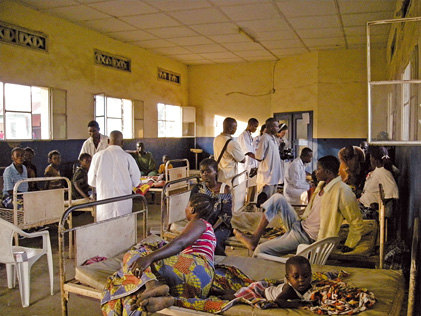
Kwamouth Ward in the DRC.
A good illustration of these collaborations is the LEAP-conducted clinical trial investigating paromomycin, an aminoglycoside antibiotic identified in the 1960's as having antileishmanial activity and having recently been approved to treat VL in India. Paromomycin could represent an improved treatment at a lower cost. With the aim to register paromomycin as a new treatment in the East African region and to have it adopted in national treatment guidelines, the study has currently treated approximately 900 patients as part of the clinical trial and a similar number of patients outside the clinical trial in 5 sites in East Africa. When patients do not qualify for enrolment in a clinical trial, they are provided treatment by the trial teams according to guidelines.
A notable step forward in multi-centred collaborative partnerships is the nifurtimox-eflornithine co-administration trial (NECT) a multi-centre clinical study to test a simplified combination of nifurtimox and eflornithine for stage 2 HAT (see Newsletter No14). The study objective is to demonstrate that the co-administration is as safe and efficacious as standard eflornithine monotherapy, but easier to use as the number of slow, intravenous infusions of nifurtimox is reduced from 54 to 16. DNDi's activities have mainly focused on the implementation of this study at three sites in DRC and have included sustainable capacity strengthening for clinical research in terms of rehabilitation and equipment. All of the 280 patients have been enrolled, the 18-month follow-up is soon to be concluded, and full efficacy and safety analysis will be completed by the end of this year.
The LEAP and HAT Platforms are a forum comprising of partners with different but complementary backgrounds: physicians, parasitologists, ethicists, and pharmacologists. These partners share one common objective, to come together and operate in a coherent manner to ultimately bring these clinical trials to term in a timely and professional manner. They should all agree on a common protocol and the pertinent strategies to follow in conducting clinical trials.
DNDi and its partners in the LEAP and HAT Platforms aim to perform high quality clinical trials whilst strictly observing ethical principles. The Ethical committee's primary role is to protect the rights of patients or volunteers participating in a clinical trial. They do this by providing a careful assessment of the trial and by providing patients with unbiased and necessary information in an easily digested manner. Along with other players like TDR, Swiss Tropical Institute, the platforms organise training on ethics in clinical trials for ethics committee members and investigators alike.
If a treatment is found to be safe end efficacious in clinical trial(s), the new treatment must then be registered in order for it to be incorporated into national treatment guidelines and ultimately become available to patients. The regional research platforms aim to facilitate the registration process by engaging local and national health authorities early on in the process, so as to ensure that needs-adapted treatments are made available to patients in as timely a manner as possible.

Arba Minch, patients waiting to be attended - May 2006, Ethiopia
Building an environment conducive to efficient clinical trials
Because infrastructure is very limited in many endemic areas, DNDi is in the process of, or has already upgraded ward and laboratory facilities in all of the clinical trial sites to enable them to meet GCP standards. For example, in Gondar, Ethiopia, a leishmaniasis Research and Treatment Centre has recently been inaugurated (see box; photo). Previously in Gondar there was insufficient room in the hospital; the VL ward functioned under a tent.
The communication challenges that present the remote locations of many of the sites are evolving. With the increased use of mobile phones and internet access, the ability to communicate with the clinical trial sites is becoming easier. For example, site investigators from Sudan no longer have to drive to the nearest town to make a phone call or access the internet. However, some HAT sites are still suffering from lack of electricity and are not covered by mobile phones.
In addition to physical infrastructure, trained staff is essential in order to carry out clinical trials according to GCP. Training is not just important at the start of a trial, but is a continuous process to update existing staff and to train new members. Frequent training topics include GCP and trial-specific procedures. From external consultants to the experienced trial site staff, the sharing of better practice principles can also act to motivate teams working in difficult field conditions.
Ensuring that all sites have standard operating procedures (SOPs) and endeavouring to standardise them across the different sites (local regulations permitting), as well as ensuring that everyone understands the need to follow SOPs and meet GCPs through a comprehensive monitoring plan is vital.
Independant clinical monitors visit trial sites on a regular basis to ensure that they are complying with good clinical and laboratory practices and SOPs They also address adverse reaction detection, reporting mechanisms, and ethical issues during planning and conducting of the trial. In addition, an independent external GCP auditor ensures that all aspects of regulatory and ethical documentation in the clinical trial comply with GCP standards. This monitoring and auditing further educate staff and reinforce the importance of carrying out clinical trials to international standards, as a positive outcome for the clinical trial will hopefully result in a new treatment.
Taking steps together in the right direction
At a symposium held during the RSTMH Centenary Meeting in London in September 2007, Prof. Ahmed El-Hassan, a Sudanese leishmaniasis expert, raised the issue about just what a change in infrastructure can accomplish.
"The project has brought African scientists in the region together to tackle a disease that knows no political boundaries: an example par excellence of South-South collaboration, about which we talk a lot and do very little... In leaps and bounds, DNDi has moved forward with activities in the region. When we started, we held our clinics under the shade of trees in protection from a very hot sun. Now, we have modern facilities that allow us to serve the unprivileged and marginalized communities with state-of-the-art medicines."
This is precisely why DNDi-sponsored LEAP and HAT Platforms will carry on with these important endeavours.
Currently in Ethiopia, 40 VL / kala-azar foci have been reported on. As Dr. Sisay Yifru, Gondar's Principle Site Investigator, puts it: "the estimate of the annual burden in the country ranges from 4,000 to 5,000 cases and is distributed throughout the Metema and Humera lowlands in the West of the country as well as the Segen Valley and its surrounding area in Konso, in the South West. Furthermore, new foci like Libokemekem in South Gondar are also becoming more and more exposed to kala-azar.
DNDi, together with its regional partners (Gondar and Addis Ababa University), is conducting clinical trials in Ethiopia. One of the trial sites initially identified was based in Gondar. However, the infrastructure to support quality clinical studies was missing to begin with. So DNDi also provided the necessary support to the Ministry of Health in strengthening the capacity of local health infrastructure by establishing a well functioning leishmaniasis Research and Treatment Centre at the University of Gondar. The team inaugurated the new treatment centre on 11 May 2008 and it is now fully operational with 24 beds. It will provide treatment to up to 300 patients per year, be they adults or children suffering from VL.
Funding for the construction came from Medicor Foundation and the Canton of Geneva with on-going funding from DFID; MSF; Canton de Genève; Region of Tuscany, Italy; MAEE France; Medicor Foundation; and Kings College for the Paromomycin trial and the LEAP Platform.
