 |
|
|
|
|


Drug discovery is very much a 'numbers game'. To give a couple of examples:
- The chemical space inhabited by most drugs could be described as relatively small molecules (less than 500 Daltons) which contain some of the following elements: carbon, oxygen, phosphorus, sulfur, nitrogen, chlorine, fluorine, bromine and less than 30 hydrogen atoms. It has been calculated that there are theoretically 1062 possible molecules fitting this description. Put another way, if one made just a single unit of each of these molecules, their combined mass would be equal to that of 100,000,000 suns.
- To be effective as a drug, the molecule must interact specifically with biological systems. However, most randomly synthesised molecules have no impact at all on living systems. In fact, all of the molecules that do interact with life, as far as we know, exist in the thin biosphere that surrounds the earth not just one copy of each molecule, but all of them.
Therefore to effectively design a new drug, some very clever strategies are necessary to improve the odds. Drugs are based around molecular interactions in living organisms through:
- Use of naturally occurring compounds such as microbial or plant toxins in the case of antibiotics.
- Use of synthetic variants of naturally occurring compounds.
- Identification of new synthetic compounds which interfere with natural molecular interactions.
For infectious diseases, the traditional approach to drug discovery is to identify a compound that selectively kills the parasite (a drug lead) and then manipulate the chemical structure until it is an effective drug (the process of lead optimisation). It is extremely rare that a drug lead could be considered as a drug without the need for optimisation because a drug must have many features in addition to being able to kill the parasite. For example, if a drug is to be taken orally it must:
- Survive in the acidic environment of the stomach.
- Survive in the intestine where bacteria and human enzymes attack large molecules.
- Cross from inside the intestine into the maze of tiny blood vessels behind it. This is a natural barrier designed to allow the flow of nutrients and exclude other molecules.
To further challenge the drug, these tiny blood vessels behind the intestine fuse into a single large blood vessel, the hepatic portal vein, which flows directly to the liver. This is the body's next line of defense as the organ contains a variety of enzymes that degrade foreign molecules or modify them so that the body can quickly excrete them.
Once past the liver, the drug must get to the parasite. It is of no use if the drug is trapped in the blood stream and the parasite resides in the central nervous system.
After all of that, the drug must pass through the kidneys, which excrete foreign molecules, and onto the liver again.
As well as passing all these barriers, the drug must be a potent poison to the parasite but relatively harmless to its human host.
The sciences associated with the different drug-like features of a molecule can be broadly classified as ADME/TOX:
A- Absorption getting into the blood
D- Distribution getting into the parasite
M- Metabolism being degraded by enzymes in the liver or other organs
E- Excretion being removed from the body
Tox- Toxicology killing the parasite and not the human
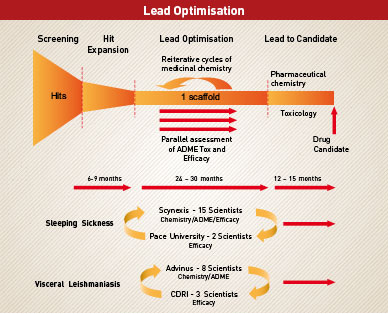
Lead optimisation is the process of taking a molecule, which kills the parasite, and changing different parts of it so that it can satisfy all of the ADME/Tox parameters necessary for it to be an effective drug, all the while maintaining the ability to kill the parasite. It is a process of trial and error coupled with a knowledge of what changes will destroy drug-like properties and what changes will make a molecule more drug-like. It involves an iterative process of changing and testing until the molecule becomes a drug candidate or researches realise that it is simply not possible to incorporate all of the necessary features into that class of molecule.
The DNDi approach is to piggyback' off research already conducted in Universities and at pharmaceutical companies by requesting access to compounds which have already shown some biological activity. In particular, we look for compounds which kill the parasites causing our target diseases without killing human cells.
Ideally, we would like to identify existing drugs which have been developed for other diseases and which are also effective against DNDi's target diseases. In some instances, clever detective work had identified drugs such as miltefosine for leishmaniasis and eflornithine for African trypanosomiasis. Both were originally developed as treatments for cancer. DNDi has other drugs in the pipeline which may be effective for treatment of trypanosomiasis or leishmaniasis. While these are our most attractive candidates in the sense of being closest to the patient, it is essential that we keep our drug pipeline full. To do so, we must move further upstream in the drug discovery process to identify new leads and to optimise them as drug candidates.
Partnerships for Lead Optimisation
The process of drug discovery and development has been likened to that of building a jet airliner. No single person has all of the skills, and the final product must be safe for millions of people effective partnerships are essential.
DNDi has had to build effective partnerships between scientists from a number of different disciplines to adequately address all of the skills necessary for lead optimisation. In particular, we have set up dedicated lead optimisation consortia for sleeping sickness and visceral leishmaniasis and plan to initiate a third partnership for Chagas' disease in the second half of 2008.
To maintain the capacity to optimise one lead series at all times, each team is comprised of 5-6 chemists, 2-3 pharmacologists and a dedicated screening facility to assess potency and efficacy. The entire infrastructure to support medicinal chemistry, in vitro and in vivo ADME/Tox and efficacy studies, is guaranteed. In addition, DNDi works with consultants who are expert in different disciplines to provide ongoing critical review of the programmes.
To produce a new drug candidate as efficiently as possible, it is very important that all players are focused on the lead optimisation programme and not distracted by other research efforts. To that end:
- Once a lead is introduced into a lead optimisation programme, it enters a critical path which promises development through to patient access unless the compound series fails because structural liabilities prevent optimisation as a drug candidate.
- Sufficient resources are allocated to guarantee the rapid turnaround of data necessary to support the medicinal chemistry effort.
- An active programme of identifying new leads is managed separately from the lead optimisation programme to ensure a constant supply of new leads.
Finally, as for all successful partnerships, effective communication and cross fertilisation is essential. Within each programme we employ a secure web based data management system to ensure that all players have real time access to all data as it is generated at the different sites. On a broader level we encourage collaboration between the teams and also work closely with other research consortia working in drug discovery for neglected diseases such as the Kinetoplastid drug-discovery research consortia at the Universities of North Carolina (Consortium for Parasitic Drug Discovery) and Dundee (Drug Discovery @ Dundee) and with other Public-Private Partnerships such as the Medicines for Malaria Venture and the TB Alliance.
Sleeping Sickness Consortium
The two lead players in DNDi's HAT Lead Optimisation Consortium are based at Research Triangle Park in North Carolina and Manhattan, New York. Scynexis Inc and Pace University bring together most of the skills required for early drug discovery.
Scynexis is a biotechnology company which provides medicinal chemistry services coupled with considerable ADME research capacity. DNDi chose to work with the company because of its track record in partnership with many of the world's major pharma companies and its commitment to neglected disease research at a senior management level. In particular, CEO Yves Ribeill has had a long term commitment which includes service on scientific advisory committees to both the World Health Organisation and Medicines for Malaria Venture in the area of drug discovery for neglected diseases. A team of fifteen Scynexis scientists including chemists and biologists, led by Dr Bob Jacobs, are dedicated to the sleeping sickness programme.
Scynexis does not have animal research capacity and Dr Cy Bacchi's team at Pace University provides the disease models in which new leads are tested. Dr Bacchi is the person behind development of the anti cancer drug, eflornithine, for sleeping sickness. Dubbed the resurrection drug, eflornithine is very effective in treating patients with stage two disease but has the disadvantage that it must be administered as an intravenous infusion in quite sophisticated clinical settings which are out of reach for many of the patients.
In addition to the two main players, DNDi commissions specialised contract research from other companies when necessary. All together, these scientists will pursue two lead optimisation programmes.
The aim of the consortium is to develop new drug candidates for sleeping sickness which are more closely aligned to the needs of the patients in poorly serviced disease endemic regions. The programme is partially funded by the Bill and Melinda Gates Foundation and it is our goal to have one compound in phase I clinical trials in 2012 and one backup compound ready to enter clinical trials if the first one fails in the clinic.
Leishmaniasis Consortium
Our visceral leishmaniasis Lead Optimisation Consortium works along similar lines to those for sleeping sickness.
In this case, we have chosen to work with two primary partners in India (Advinus Therapeutics and the Central Drug Research Institute) where the disease is endemic.
Advinus Therapeutics will provide medicinal chemistry services together with ADME/Tox resources, and 8 scientists within the company are currently devoted to the programme. Dr Rashmi Barbhaiya, CEO of Advinus Therapeutics, has a strong commitment to neglected disease research and has been involved with drug discovery programmes for new antimalarial drug candidates. As head and cofounder of one of India's largest biotechnology companies, Dr Barbhaiya has transmitted his enthusiasm to the company's major financial backers and gained their support to commit company resources to neglected disease drug-discovery research.
The Central Drug Research Institute, based in Lucknow, is a publicly funded institute with a proven track record in anti-parasitic drug discovery, having already developed and registered Arteether and Bulaquin as antimalarial drugs for the Indian market. A team led by Dr S K Puri will support the DNDi programme by parasitological screening of leads developed by the Scynexis team.
Also partially funded by the Bill and Melinda Gates Foundation, the visceral leishmaniasis team aims to have one new drug candidate in phase I clinical trials by 2013.