 |
|
|
|
|


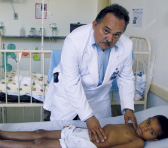
Ninety per cent of VL cases in Latin America (LA) are estimated to occur in Brazil, most of them in children. In 2007, Brazil reported some 3,500 new cases and a fatality rate of 5.4%, which indicates an improving trend compared to previous years, though there is evidence indicating around 40% underestimation of numbers of VL cases and deaths in the epidemiological surveillance system(1). In the 1980s-1990s, VL was mainly confined to rural areas of the NE region of Brazil, but over the last decade it has reached other regions as well as urban centres. The disease is a zoonosis in LA, which requires additional efforts from governments to control not only the phlebotomine vector, but also the vast population of dogs that host the parasite(1). The human cases, caused by Leishmania (L.) chagasi, are treated with highly toxic, long-course parenteral treatments that require either hospitalisation or daily visits to the clinic.
“Due to epidemiological characteristics and lack of a complete understanding of the multiple elements that comprise the transmission chain of VL, the control strategies have not achieved the expected efficacy. VL control in large urban areas, reduction of lethality and VL-HIV co-infection are among major challenges faced by the control programme. By investing in technologies for new diagnostics tools and new therapies for prevention and control, together with improvement on to access for diagnosis/ treatment and broadening the use of liposomal amphotericin B, we aim to achieve the expected impact on VL control in Brazil” comments Gerson Penna, Secretary of Health Surveillance - Brazilian Ministry of Health.
In response to this demanding situation, DNDi, with support from the Brazilian Ministry of Health, is fostering a multicentric study to evaluate the safety and efficacy of different drug combinations to treat VL patients in Brazil. In the coming months, DNDi, in partnership with Instituto René Rachou (IRR), Fiocruz, and Universidade de Brasília, will identify and select the participating centres, develop clinical protocols, provide GCP training, and strengthen capacity according to the centres’ needs. The study will be fully implemented in 2010. With strong support from DNDi, IRR is also becoming a reference centre in Latin America for in vitro and in vivo drug screening for leishmaniasis. Activity against different species of CL (cutaneous leishmaniasis) and VL is evaluated, aiming to identify new promising compounds that can be translated into new therapies.