 | |

Filling the gaps
in HAT drug development
in HAT drug development
Currently, few drugs exist for the treatment of either Stage 1 or Stage 2 human African trypanosomiasis (HAT), and severe toxic side effects are a common problem (see Nect Project). The difficulty of diagnosis, stage determination, and increasing numbers of treatment failures pose additional clinical challenges. The overall strategy of DNDi is to build a dynamic portfolio to fill the numerous gaps in the drug pipeline that have led to few drugs developed for HAT patients. In fact, the last two drugs developed for HAT were melarsoprol (1947) and eflornithine (introduced in the 1980s). The only recent new drug development is the new diamidine, DB289, by a University of North-Carolina-led consortium supported by the Bill & Melinda Gates Foundation, which is currently in phase III clinical trials for stage 1 HAT.
DNDi is currently involved with partners in a multi-centre clinical study to test a simplified combination of nifurtimox and eflornithine (NECT) for stage 2 HAT (see Nect Project). The objective is to demonstrate that the combination is as effective and safe as the standard eflornithine monotherapy, but more easy to use. The study pools data from 6 sites in the Democratic Republic of Congo, Congo-Brazzaville and Uganda.
 While this combination is expected to represent an improved treatment for patients, it is not ideal because of a still rather complicated administration and the need for close patient follow-up. Hence, major efforts are needed to bring truly innovative drugs into the pipeline. As a consequence, the majority of the 12 projects in the current DNDi HAT portfolio are in the discovery stage (see Current HAT portfolio diagram).
While this combination is expected to represent an improved treatment for patients, it is not ideal because of a still rather complicated administration and the need for close patient follow-up. Hence, major efforts are needed to bring truly innovative drugs into the pipeline. As a consequence, the majority of the 12 projects in the current DNDi HAT portfolio are in the discovery stage (see Current HAT portfolio diagram).
By focusing on discovery today, DNDi aims to ensure that a first promising, novel drug candidate enters into preclinical development by 2008, with several other opportunities lined up behind, and to build an effective virtual R&D network with:
• Links to academic discovery groups, e.g., for trypanothione reductase inhibitors and cysteine protease inhibitors (see Discovery and preclinical projects)
• Access to chemical diversity – expand identification of novel pharmacophores from selected libraries, e.g Kitasato Institute library (see Discovery and preclinical projects)
• Proactive approach on compounds with known antiparasitic activities – e.g., ascofuranone and nitroimidazoles (see Discovery and preclinical projects)
This effort is expected to deliver 2 new drug candidates for clinical testing by the end of 2011 (possibly one drug earlier).
To ensure that future drug candidates can be tested in a timely manner, DNDi has catalysed a network involving the HAT control programmes and researchers from the key endemic countries to strenghten their clinical trial capacity and identify and overcome possible regulatory, logistic and other practical hurdles (see Special section).
DNDi is currently involved with partners in a multi-centre clinical study to test a simplified combination of nifurtimox and eflornithine (NECT) for stage 2 HAT (see Nect Project). The objective is to demonstrate that the combination is as effective and safe as the standard eflornithine monotherapy, but more easy to use. The study pools data from 6 sites in the Democratic Republic of Congo, Congo-Brazzaville and Uganda.
 While this combination is expected to represent an improved treatment for patients, it is not ideal because of a still rather complicated administration and the need for close patient follow-up. Hence, major efforts are needed to bring truly innovative drugs into the pipeline. As a consequence, the majority of the 12 projects in the current DNDi HAT portfolio are in the discovery stage (see Current HAT portfolio diagram).
While this combination is expected to represent an improved treatment for patients, it is not ideal because of a still rather complicated administration and the need for close patient follow-up. Hence, major efforts are needed to bring truly innovative drugs into the pipeline. As a consequence, the majority of the 12 projects in the current DNDi HAT portfolio are in the discovery stage (see Current HAT portfolio diagram).By focusing on discovery today, DNDi aims to ensure that a first promising, novel drug candidate enters into preclinical development by 2008, with several other opportunities lined up behind, and to build an effective virtual R&D network with:
• Links to academic discovery groups, e.g., for trypanothione reductase inhibitors and cysteine protease inhibitors (see Discovery and preclinical projects)
• Access to chemical diversity – expand identification of novel pharmacophores from selected libraries, e.g Kitasato Institute library (see Discovery and preclinical projects)
• Proactive approach on compounds with known antiparasitic activities – e.g., ascofuranone and nitroimidazoles (see Discovery and preclinical projects)
This effort is expected to deliver 2 new drug candidates for clinical testing by the end of 2011 (possibly one drug earlier).
To ensure that future drug candidates can be tested in a timely manner, DNDi has catalysed a network involving the HAT control programmes and researchers from the key endemic countries to strenghten their clinical trial capacity and identify and overcome possible regulatory, logistic and other practical hurdles (see Special section).
Distribution of T.b. gambiense and T.b. rhodesiense sleeping sickness
in sub-Saharan Africa
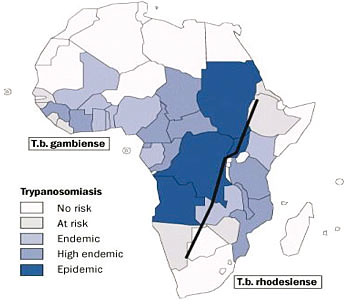
in sub-Saharan Africa

The challenge of the blood brain barrier
The blood-brain barrier (BBB) is a membrane that controls the passage of substances from the blood into the central nervous system (CNS). A major challenge in the treatment of human African trypanosomiasis is to find therapeutic agents that can cross this barrier to eliminate parasites in the brain. In its neuroprotective role, the BBB functions to hinder the entry of most substances to the fragile brain.
The blood-brain barrier (BBB) is a membrane that controls the passage of substances from the blood into the central nervous system (CNS). A major challenge in the treatment of human African trypanosomiasis is to find therapeutic agents that can cross this barrier to eliminate parasites in the brain. In its neuroprotective role, the BBB functions to hinder the entry of most substances to the fragile brain.
DNDi objectives to deliver new HAT drugs over the next decade:
• In 2008
– 1 new drug candidates in preclinical development
– An effective, simpler and safer first-line treatment for stage 2 HAT available for the patients (eflornithine-nifurtimox)
• By the end of 2011
– 2 new drug candidates in clinical development
– 2 back-up compounds (preclinical development done)
• In 2014
– At least 1 new drug available to patients
– Back-up candidates in the pipeline
• In 2008
– 1 new drug candidates in preclinical development
– An effective, simpler and safer first-line treatment for stage 2 HAT available for the patients (eflornithine-nifurtimox)
• By the end of 2011
– 2 new drug candidates in clinical development
– 2 back-up compounds (preclinical development done)
• In 2014
– At least 1 new drug available to patients
– Back-up candidates in the pipeline
Published by Drugs for Neglected Diseases Initiative - 1 Place St Gervais 1201 Geneva Switzerland - Photo credits: DNDi
Editor: Ann-Marie Sevcsik - Tel: +41 22 906 9230 - Fax: +41 22 906 9231 - www.dndi.org
Editor: Ann-Marie Sevcsik - Tel: +41 22 906 9230 - Fax: +41 22 906 9231 - www.dndi.org