New anti-malarial – AS/AQ – to patients by 2006 In April, Sanofi-Aventis signed an agreement with DNDi to complete the development of the anti malarial artesunate-amodiaquine, and begin the registration process in Morocco by November 2005. The news that a new drug will be available to patients suffering from malaria by the end of 2006 received significant coverage in the media.  New member on Board Morten Rostrup, former President of MSF International leaves the DNDi Board after two years of excellent guidance and direction. We will miss his wisdom and insights at our meetings.
Always quick to remind us that the neglected patient was most important to DNDi's work, Morten believed that "the process of developing new drugs must mirror the urgent need to find adequate treatment for neglected diseases. This will make it ethically necessary for DNDi to take some risks, which in the end will be to the benefit of our patients."
In Morten's place, DNDi warmly welcomes Rowan Gillies, current President of MSF International, who also brings to the table his experience treating neglected patients in Afghanistan, Sierra Leone, Sudan, Democratic Republic of Congo, and Liberia as a field doctor for MSF. | | New collaborations for DNDi: Kitasato and CDRI DNDi has signed an agreement in July with Kitasato Institute, Japan, to conduct screening of compounds effective against the parasite that causes human African trypanosomiasis, commonly known as sleeping sickness. In a similar move, DNDi will sign an agreement with the Central Drug Research Institute in Lucknow, India, to screen compounds for sleeping sickness and kala-azar.  More proposals from disease endemic countries for portfolio Forty eight project proposals were submitted to the call for letters early this year, a greater proportion of which were received from scientific groups in disease endemic regions (see figure). The Scientific Advisory Committee has invited full proposals from 6 principal investigators - 3 discovery projects, 2 clinical development projects, and one model development project. The full proposals will be reviewed in October and final recommendations presented to the Board in December 2005. 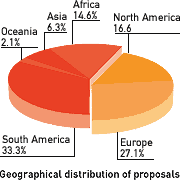 |




